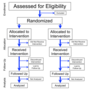
Clinical trials are prospective biomedical or behavioral research studies on human participants designed to answer specific questions about biomedical...
112 KB (12,658 words) - 10:41, 24 April 2024
expressed specifically, a clinical trial phase is capitalized both in name and Roman numeral, such as "Phase I" clinical trial. If the drug successfully...
32 KB (3,806 words) - 20:17, 29 May 2024
factors not under direct experimental control. Examples of RCTs are clinical trials that compare the effects of drugs, surgical techniques, medical devices...
88 KB (9,887 words) - 00:24, 31 May 2024
ClinicalTrials.gov is a registry of clinical trials. It is run by the United States National Library of Medicine (NLM) at the National Institutes of Health...
19 KB (1,865 words) - 18:50, 13 December 2023
A pragmatic clinical trial (PCT), sometimes called a practical clinical trial (PCT), is a clinical trial that focuses on correlation between treatments...
8 KB (974 words) - 15:21, 26 November 2023
remains a viable treatment option. Similar trials have been done with chondroitin. In general, the clinical trials of the mid-1990s that furnished positive...
45 KB (5,595 words) - 11:52, 29 May 2024
Preregistration (science) (redirect from Clinical trial registration)
methods, and/or analyses of a scientific study before it is conducted. Clinical trial registration is similar, although it may not require the registration...
56 KB (5,841 words) - 08:21, 9 June 2024
regulation(s) addressing the conduct of clinical trials of investigational products of the jurisdiction where trial is conducted. (ICH E6) Approval (in relation...
78 KB (11,571 words) - 05:27, 12 May 2024
A Clinical Trial Management System (CTMS) is a software system used by biotechnology and pharmaceutical industries to manage clinical trials in clinical...
8 KB (866 words) - 12:06, 7 May 2024
clinical research trials. The term may also refer to any disease or sign that strongly motivates withdrawal of an individual or entity from the trial...
15 KB (1,932 words) - 20:33, 24 April 2024
PMC 4497427. PMID 26175929. Clinical trial number NCT02704858 for "Safety and Efficacy Study in Recurrent Grade IV Glioma" at ClinicalTrials.gov Wikimedia Commons...
80 KB (8,685 words) - 05:40, 9 June 2024
An in silico clinical trial, also known as a virtual clinical trial, is an individualized computer simulation used in the development or regulatory evaluation...
21 KB (2,457 words) - 07:54, 6 May 2024
Preclinical development (redirect from Pre-clinical trial)
or nonclinical studies) is a stage of research that begins before clinical trials (testing in humans) and during which important feasibility, iterative...
7 KB (804 words) - 14:26, 27 January 2024
Stratification of clinical trials is the partitioning of subjects and results by a factor other than the treatment given. Stratification can be used to...
3 KB (354 words) - 12:52, 22 September 2021
MEK inhibitor (section In clinical trials)
melanoma with a BRAF V600E or V600K mutation. Selumetinib, had a phase 2 clinical trial for non-small cell lung cancer (NSCLC) which demonstrated an improvement...
6 KB (719 words) - 23:12, 12 May 2024
Parexel (section TGN1412 clinical trial)
It conducts clinical trials on behalf of its pharmaceutical clients to expedite the drug approval process. It is the second largest clinical research organization...
28 KB (2,619 words) - 10:38, 6 June 2024
Controlled clinical trial may refer to: Clinical trial, the medical research technique Contemporary Clinical Trials, the continuation of the discontinued...
235 bytes (57 words) - 00:59, 15 July 2017
name Ozempic. Clinical trials started in January 2016 and ended in May 2017. In March 2021, in a phase III randomized, double-blind, trial, 1,961 adults...
66 KB (5,418 words) - 18:14, 27 May 2024
includes conducting interventional studies (clinical trials) or observational studies on human participants. Clinical research can cover any medical method...
9 KB (989 words) - 21:52, 9 March 2024
Hawthorne effect (redirect from Clinical trial effect)
Various medical scientists have studied possible trial effect (clinical trial effect) in clinical trials. Some postulate that, beyond just attention and...
34 KB (3,904 words) - 02:03, 10 June 2024
Good clinical practice (GCP) is an international quality standard, which governments can then transpose into regulations for clinical trials involving...
6 KB (677 words) - 09:17, 15 May 2024
Clinical trials are often assigned contrived acronyms. Some common themes include acronyms excluding words from the acronym and including letters taken...
7 KB (810 words) - 00:43, 2 May 2023
An academic clinical trial is a clinical trial not funded by pharmaceutical or biotechnology company for commercial ends but by public-good agencies (usually...
3 KB (375 words) - 22:22, 5 January 2022
Clinical trials in India refers to clinical research in India in which researchers test drugs and other treatments on research participants. NDCTR 2019...
47 KB (5,533 words) - 01:19, 16 December 2023
An open-label trial, or open trial, is a type of clinical trial in which information is not withheld from trial participants. In particular, both the...
3 KB (284 words) - 18:11, 14 March 2023
Contemporary Clinical Trials is a monthly peer-reviewed medical journal covering clinical trials and research design in clinical medicine. It was established...
2 KB (86 words) - 21:50, 17 March 2024
Placebo-controlled study (redirect from Placebo-controlled clinical study)
or no treatment at all?" As one early clinical trial researcher wrote, "the first object of a therapeutic trial is to discover whether the patients who...
32 KB (4,319 words) - 01:32, 11 February 2024
Risankizumab (section Clinical trials)
primarily from five clinical trials (Trial 1/NCT02684370, Trial 2/NCT02684357, Trial 3/NCT02672852, Trial 4/ NCT02694523, and Trial 5/NCT02054481) of 1606...
13 KB (969 words) - 19:29, 20 December 2023
Clinical study design is the formulation of trials and experiments, as well as observational studies in medical, clinical and other types of research...
10 KB (1,030 words) - 17:15, 26 February 2024
Riociguat (section Phase I clinical trials)
hypertension in combination with idiopathic interstitial pneumonia (PH-IIP). A clinical trial testing riociguat for this purpose was prematurely terminated because...
20 KB (2,115 words) - 04:18, 30 May 2024