effect, change in electron density Asymmetric induction, preferring one stereoisomer over another Grammar induction Inductive bias Inductive probability... 2 KB (196 words) - 16:52, 29 June 2023 |
 | Enantioselective synthesis (redirect from Asymmetric catalyst) through interactions at the transition state. This biasing is known as asymmetric induction and can involve chiral features in the substrate, reagent, catalyst... 41 KB (4,724 words) - 12:46, 31 March 2024 |
 | Ene reaction (section Internal asymmetric induction) carbonyl groups. The catalysts were found to afford high levels of asymmetric induction in several processes, including the ene reaction of ethyl glyoxylate... 27 KB (3,335 words) - 00:18, 25 November 2023 |
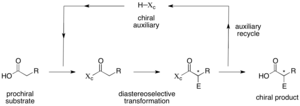 | Chiral auxiliary (redirect from Myers' asymmetric alkylation) "Preparation of an Optically Active Prostaglandin Intermediate via Asymmetric Induction". J. Am. Chem. Soc. 97 (23): 6908–6909. doi:10.1021/ja00856a074.... 34 KB (3,692 words) - 07:36, 24 August 2023 |
C2-Symmetric ligands (redirect from Asymmetric ligand) used in Sharpless asymmetric dihydroxylation While the presence of any symmetry element within a ligand intended for asymmetric induction might appear counterintuitive... 11 KB (1,164 words) - 21:28, 31 March 2024 |
Sharpless asymmetric dihydroxylation (also called the Sharpless bishydroxylation) is the chemical reaction of an alkene with osmium tetroxide in the presence... 20 KB (2,175 words) - 13:53, 30 November 2023 |
1,3-Dipolar cycloaddition (section Stereoselectivity and asymmetric induction of the 1,3-dipolar cycloaddition reaction mediated by metal catalysis of diazocarbonyl compounds) in particular Rh2[(S)-DOSP]4 and Rh2[(S)-BPTV]4 can induce modest asymmetric induction and was used to synthesize the antifungal agent pseudolaric acid... 65 KB (6,426 words) - 14:32, 7 March 2024 |
Asymmetric hydrogenation is a chemical reaction that adds two atoms of hydrogen to a target (substrate) molecule with three-dimensional spatial selectivity... 64 KB (6,351 words) - 14:54, 11 December 2023 |
Tsuji–Trost reaction (redirect from Trost asymmetric allylic alkylation) Asymmetric induction in allylic alkylations." J. Am. Chem. Soc. 1973, 95, 8200–8201. doi:10.1021/ja00805a056. Trost, B. M.; Strege, P. E. "Asymmetric... 24 KB (2,771 words) - 01:05, 8 March 2024 |
= OR*). (1) Asymmetric versions of the above reaction have taken advantage of a number of strategies for achieving asymmetric induction. The highest... 14 KB (1,634 words) - 16:11, 7 March 2021 |
structure. He also did work in stereochemistry and Cram's rule of asymmetric induction is named after him. In 1973, Cram collaborated on research with Irish... 16 KB (1,320 words) - 09:39, 7 April 2024 |
employing chiral starting materials have been used to effect either asymmetric induction or simple diastereoselection (1) After carbanion formation, the [2... 13 KB (1,449 words) - 18:59, 23 January 2024 |
 | predictable stereodirecting effects resulting in high levels of asymmetric induction. Racemization of the newly created carbon-nitrogen stereo center... 15 KB (1,583 words) - 03:48, 22 October 2022 |
transfer of an H2 molecule from the reductant to the substrate. Asymmetric induction in transition metal catalyzed reactions is achieved through the use... 12 KB (1,406 words) - 21:04, 19 March 2024 |
rearrangement Coupling reaction Crabbé reaction Craig method Cram's rule of asymmetric induction Creighton process Criegee reaction Criegee rearrangement Cross metathesis... 38 KB (3,433 words) - 17:07, 5 January 2024 |
insertions are seen. Both inter- and intra-molecular insertions admit asymmetric induction from a chiral metal catalyst. Carbenes can form adducts with nucleophiles... 22 KB (2,400 words) - 04:37, 4 February 2024 |
(3 January 2014). "Foldamer-Mediated Remote Stereocontrol: >1,60 Asymmetric Induction" (PDF). Angewandte Chemie International Edition. 53 (1): 151–155... 5 KB (352 words) - 09:47, 7 April 2024 |
Chiral Lewis acid (section Asymmetric synthesis) one enantiomer or diastereomer over the other is formally known as asymmetric induction. In this kind of Lewis acid, the electron-accepting atom is typically... 11 KB (1,242 words) - 23:24, 12 November 2023 |
Etter, Jeffrey B. (1987). "Lanthanides in organic synthesis. 8. 1.3-Asymmetric induction in intramolecular Reformatskii-type reactions promoted by samarium... 13 KB (1,269 words) - 19:55, 31 October 2023 |
 | Organolithium reagent (section Asymmetric metalation) organolithium species can be obtained through asymmetric metalation of prochiral substrates. Asymmetric induction requires the presence of a chiral ligand... 55 KB (5,971 words) - 08:36, 6 March 2024 |
 | et al. (1981). "Asymmetric total synthesis of erythromcin. 1. Synthesis of an erythronolide A secoacid derivative via asymmetric induction". J. Am. Chem... 17 KB (1,720 words) - 20:13, 14 April 2024 |
 | chiral chemical synthesis, and specifically of the phenomenon of asymmetric induction during nucleophilic attack at hindered carbonyl centers (see the... 15 KB (1,573 words) - 19:00, 8 July 2023 |
 | discrimination between these possible enantiomers that requires asymmetric induction. Torquoselectivity is also used to describe selective electrocyclic... 4 KB (373 words) - 09:24, 5 January 2023 |
 | bulky tosyl group on the imine electrophile is responsible for the asymmetric induction taking place. In another application DOM is applied in placing a... 9 KB (1,041 words) - 15:17, 1 February 2024 |






