 | Pfizer–BioNTech's BNT162b2 COVID-19 vaccine for widespread use. On 11 December, the FDA gave emergency use authorization for the Pfizer–BioNTech COVID-19... 78 KB (7,926 words) - 15:19, 26 April 2024 |
 | Operation Warp Speed (section Pfizer–BioNTech) at BioNTech's laboratories in Mainz, Germany, just days after the SARS-Cov-2 genetic sequence was first made public. In September 2020, BioNTech received... 72 KB (5,994 words) - 16:34, 28 April 2024 |
regulatory authority recognized by the World Health Organization (WHO): Pfizer–BioNTech, Oxford–AstraZeneca, Sinopharm BIBP, Moderna, Janssen, CoronaVac, Covaxin... 318 KB (19,351 words) - 20:00, 21 April 2024 |
 | Katalin Karikó (category BioNTech) associated with BioNTech RNA Pharmaceuticals, first as a vice president and promoted to senior vice president in 2019. In 2022, she left BioNTech to devote... 38 KB (4,177 words) - 12:44, 18 April 2024 |
 | Uğur Şahin (category BioNTech) is a German oncologist and immunologist. He is the founder and CEO of BioNTech, which developed one of the major vaccines against COVID-19. His main fields... 56 KB (6,111 words) - 22:28, 5 April 2024 |
On 25 January 2021, the TGA provisionally approved the two-dose Pfizer–BioNTech COVID-19 vaccine, named COMIRNATY, for use within Australia. The provisional... 23 KB (1,797 words) - 14:29, 7 January 2024 |
 | and neurology. The company's largest products by sales are the Pfizer–BioNTech COVID-19 vaccine ($11 billion in 2023 revenues), apixaban ($6 billion in... 170 KB (14,804 words) - 16:38, 22 April 2024 |
 | Özlem Türeci (category BioNTech) company BioNTech, which in 2020 developed the first messenger RNA-based vaccine approved for use against COVID-19. Türeci has served as BioNTech's chief... 41 KB (3,474 words) - 00:55, 27 April 2024 |
 | Pfizer-BioNTech vaccine. The children under 15 years old need parental approval to be vaccinated, and they only get 1 dose of the Pfizer-BioTech vaccine... 6 KB (481 words) - 11:22, 11 April 2024 |
conditional and temporary authorization to supply for use of the Pfizer–BioNTech COVID-19 vaccine codenamed BNT162b2 (later branded as Comirnaty). This... 24 KB (2,284 words) - 11:01, 6 March 2024 |
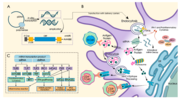
weakened immune systems. Several COVID‑19 vaccines, such as the Pfizer–BioNTech and Moderna vaccines, use RNA to stimulate an immune response. When introduced... 192 KB (21,930 words) - 19:29, 15 April 2024 |
 | PEGylated lipid is used as an excipient in both the Moderna and Pfizer–BioNTech vaccines for SARS-CoV-2. Both RNA vaccines consist of messenger RNA, or... 37 KB (3,904 words) - 15:19, 17 March 2024 |
 | COVID-19 pandemic in Bulgaria (section Total number of vaccinations (Pfizer-BioNTech + Moderna + AstraZeneca)) The COVID-19 pandemic in Bulgaria was a part of the worldwide pandemic of coronavirus disease 2019 (COVID-19) caused by severe acute respiratory syndrome... 298 KB (12,218 words) - 04:52, 8 April 2024 |
 | SARS-CoV-2 Beta variant (section Pfizer–BioNTech) African Health Products Regulatory Authority (SAHPRA) approved the Pfizer-BioNTech vaccine (BNT162b2) for section 21 Emergency Use Authorisation. On 1 April... 72 KB (6,070 words) - 06:39, 3 February 2024 |
July 2021, BioNTech's Chinese sales agent Fosun Pharma announced that Foxconn and TSMC had reached an agreement to purchase 10 million BioNTech COVID-19... 116 KB (9,744 words) - 14:53, 28 April 2024 |