Lithium peroxide is the inorganic compound with the formula Li2O2. Lithium peroxide is a white solid, and unlike most other alkali metal peroxides, it...
6 KB (527 words) - 17:20, 7 May 2024
the principal lithium mineral spodumene (LiAlSi2O6) is 8.03%. Lithium oxide forms along with small amounts of lithium peroxide when lithium metal is burned...
7 KB (480 words) - 18:59, 8 March 2024
decompose lithium peroxide into lithium ions, thereby charging the battery. During discharge, oxygen from air replenished the lithium peroxide. List of...
47 KB (5,233 words) - 12:14, 1 May 2024
useful in scuba gear, submarines, etc. Lithium peroxide and potassium superoxide have similar uses. Sodium peroxide was once used on a large scale for the...
7 KB (547 words) - 18:46, 29 September 2023
hygroscopic and are used as desiccants for gas streams. Lithium hydroxide and lithium peroxide are the salts most commonly used in confined areas, such...
135 KB (13,448 words) - 21:42, 15 May 2024
Press. ISBN 0-8493-0487-3. Khosravi J (2007). Production of Lithium Peroxide and Lithium Oxide in an Alcohol Medium. Chapter 9: Results. ISBN 978-0-494-38597-5...
14 KB (1,090 words) - 01:54, 25 April 2024
superoxide. It is currently unclear whether lithium should react analogously. Lithium oxide Lithium peroxide Andrews, Lester (1969-05-15). "Infrared Spectrum...
11 KB (1,208 words) - 20:58, 8 December 2023
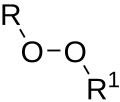
In organic chemistry, organic peroxides are organic compounds containing the peroxide functional group (R−O−O−R′). If the R′ is hydrogen, the compounds...
23 KB (2,413 words) - 16:53, 18 April 2024
Hydrogen peroxide is a chemical compound with the formula H2O2. In its pure form, it is a very pale blue liquid that is slightly more viscous than water...
91 KB (9,169 words) - 13:31, 18 April 2024
Lithium peroxide (Li2O2) is a white solid that melts at 195 °C. It reacts with carbon dioxide to form lithium carbonate and oxygen. Sodium peroxide (Na2O2)...
5 KB (457 words) - 17:49, 9 February 2024
elements. Lithium peroxide is formed upon treating lithium hydroxide with hydrogen peroxide: 2 LiOH + H2O2 → Li2O2 + 2 H2O Barium peroxide is prepared...
9 KB (1,086 words) - 20:31, 5 October 2023
Potassium peroxide is an inorganic compound with the molecular formula K2O2. It is formed as potassium reacts with oxygen in the air, along with potassium...
3 KB (169 words) - 11:03, 23 March 2023
Alkali metal (redirect from Lithium family)
many others. Lithium burns in air to form lithium oxide, but sodium reacts with oxygen to form a mixture of sodium oxide and sodium peroxide. Potassium...
216 KB (23,537 words) - 04:34, 14 May 2024
Carbon dioxide scrubber (section Lithium hydroxide)
H2O(g) Lithium peroxide can also be used as it absorbs more CO2 per unit weight with the added advantage of releasing oxygen. In recent years lithium orthosilicate...
18 KB (2,185 words) - 20:00, 25 February 2024
Lithium carbide, Li 2C 2, often known as dilithium acetylide, is a chemical compound of lithium and carbon, an acetylide. It is an intermediate compound...
8 KB (889 words) - 22:24, 11 February 2024
Peter Bruce (section Lithium-air battery)
G. (22 February 2018). "Kinetics of lithium peroxide oxidation by redox mediators and consequences for the lithium–oxygen cell". Nature Communications...
14 KB (1,397 words) - 13:25, 1 May 2024
oxide – LiCoO2 Lithium oxide – Li2O Lithium peroxide – Li2O2 Lithium hydride – LiH Lithium hydroxide – LiOH Lithium iodide – LiI Lithium iron phosphate...
119 KB (8,726 words) - 04:34, 18 April 2024
circumstances. Lithium hydroxide and particularly lithium peroxide may be used where low mass is important, such as in space stations and space suits. Lithium peroxide...
83 KB (8,523 words) - 12:35, 3 May 2024
Li2MoO4 lithium molybdate 13568–40–6 Li2O lithium oxide 12057–24–8 Li2O2 lithium peroxide 12031–80–0 Li2S lithium sulfide 12136–58–2 Li2SO4 lithium sulfate...
139 KB (120 words) - 23:26, 30 April 2024
Rubidium peroxide is rubidium's peroxide with the chemical formula Rb2O2. Rubidium peroxide can be produced by rapidly oxidizing rubidium in liquid ammonia...
2 KB (190 words) - 16:59, 10 January 2024
Hydroperoxide (category Organic peroxides)
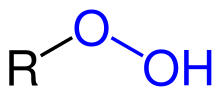
called organic hydroperoxides. Such compounds are a subset of organic peroxides, which have the formula ROOR. Organic hydroperoxides can either intentionally...
11 KB (1,149 words) - 09:21, 21 April 2024
with peroxide-containing reagents, which donate a single oxygen atom. Safety considerations weigh on these reactions because organic peroxides are prone...
22 KB (2,319 words) - 15:49, 26 April 2024
Caesium peroxide or cesium peroxide is an inorganic compound of caesium and oxygen with the chemical formula Cs2O2. It can be formed from caesium metal...
4 KB (344 words) - 22:31, 12 April 2024
Liquid rocket propellant (section Lithium and fluorine)
engine which was test fired in 2023. High-test peroxide High test peroxide is concentrated Hydrogen peroxide, with around 2% to 30% water. It decomposes...
49 KB (3,559 words) - 19:17, 7 May 2024
Li2NbO3 lithium metaniobate 12031-63-9 Li2N2O2 lithium hyponitrite Li2O lithium oxide 12057-24-8 Li2O2 lithium peroxide 12031-80-0 Li2S lithium sulfide...
182 KB (107 words) - 13:33, 17 April 2024
Battery recycling (redirect from Lithium-ion battery recycling)
chemicals). Rechargeable nickel–cadmium (Ni-Cd), nickel metal hydride (Ni-MH), lithium-ion (Li-ion) and nickel–zinc (Ni-Zn), can also be recycled. Disposable...
48 KB (4,754 words) - 01:57, 18 April 2024
perchlorate, solution UN 1471 5.1 Lithium hypochlorite, dry or Lithium hypochlorite mixtures, dry UN 1472 5.1 Lithium peroxide UN 1473 5.1 Magnesium bromate...
7 KB (159 words) - 17:33, 14 March 2022
use is not recommended in people with thyroid problems or who are taking lithium. Povidone-iodine is a chemical complex of povidone, hydrogen iodide, and...
23 KB (2,330 words) - 16:48, 11 May 2024
sodium chlorate (NaClO3), which is mixed with less than 5 percent barium peroxide (BaO2) and less than 1 percent potassium perchlorate (KClO4). The explosives...
9 KB (1,048 words) - 10:02, 16 April 2024
Ether (section Peroxide formation)
indicates presence of peroxides. The dangerous properties of ether peroxides are the reason that diethyl ether and other peroxide forming ethers like tetrahydrofuran...
19 KB (1,839 words) - 16:54, 7 May 2024