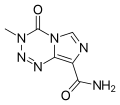
| Olaparib, sold under the brand name Lynparza, is a medication for the maintenance treatment of BRCA-mutated advanced ovarian cancer in adults. It is a... 24 KB (2,161 words) - 05:43, 7 April 2024 |
 | for long-term neurodegenerative diseases. Olaparib: In December, 2014, the EMA and US FDA approved olaparib as monotherapy (at 400 mg taken twice per... 20 KB (2,052 words) - 20:57, 21 April 2024 |
 | Fleming GF, et al. (October 2014). "Combination cediranib and olaparib versus olaparib alone for women with recurrent platinum-sensitive ovarian cancer:... 8 KB (644 words) - 19:21, 20 December 2023 |
 | for those with mutations activating the protein PIK3CA. PARP inhibitors (olaparib and talazoparib) for those with mutations that inactivate BRCA1 or BRCA2... 133 KB (14,898 words) - 19:40, 5 May 2024 |
bromide/formoterol Goserelin Isosorbide mononitrate Metoprolol Motavizumab Olaparib Omeprazole Osimertinib Palivizumab Propofol Quetiapine Ravulizumab Roflumilast... 8 KB (967 words) - 07:52, 29 October 2022 |
 | million. In December, the company received accelerated FDA approval for Olaparib in the treatment of women with advanced ovarian cancer who have a BRCA... 85 KB (7,811 words) - 07:01, 8 May 2024 |
bromide/formoterol Goserelin Isosorbide mononitrate Metoprolol Motavizumab Olaparib Omeprazole Osimertinib Palivizumab Propofol Quetiapine Ravulizumab Roflumilast... 20 KB (1,377 words) - 17:48, 12 March 2024 |