 | Pembrolizumab, sold under the brand name Keytruda, is a humanized antibody used in cancer immunotherapy that treats melanoma, lung cancer, head and neck... 72 KB (6,842 words) - 05:58, 10 April 2024 |
As adjuvant therapy, mRNA-4157 monotherapy and in combination with pembrolizumab have been investigated in patients with resected solid tumors (melanoma... 9 KB (899 words) - 20:37, 28 April 2024 |
Favezelimab/pembrolizumab is a fixed-dose combination of two monoclonal antibodies developed by Merck to treat various cancers. Timmerman, John; Lavie... 7 KB (422 words) - 20:10, 24 April 2024 |
 | Cancer immunotherapy (section Pembrolizumab) checkpoint inhibitors include antibodies such as ipilimumab, nivolumab, and pembrolizumab. Dendritic cell therapy provokes anti-tumor responses by causing dendritic... 90 KB (9,943 words) - 14:26, 10 May 2024 |
It is being tested by itself and in a fixed-dose combination with pembrolizumab. Garralda, E.; Sukari, A.; Lakhani, N. J.; Patnaik, A.; Lou, Y.; Im... 7 KB (511 words) - 20:09, 24 April 2024 |
 | liver, and that he had begun treatment with the immunotherapy drug pembrolizumab and was about to start radiation therapy. His healthcare was managed... 320 KB (28,380 words) - 20:46, 4 May 2024 |
treatment options. Pembrolizumab monotherapy is an appealing approach for these patients, with median OS of 30 months with pembrolizumab single agent compared... 59 KB (6,487 words) - 18:42, 27 December 2023 |
 | drugs or products, each with over $1 billion in revenue: Keytruda (pembrolizumab), a humanized antibody used in cancer immunotherapy that had $14.3 billion... 95 KB (9,333 words) - 08:57, 9 May 2024 |
quality-assured biosimilars Afatinib and gefitinib are alternatives Pembrolizumab is an alternative, including quality-assured biosimilars Enzalutamide... 77 KB (4,835 words) - 05:22, 23 March 2024 |
 | repair benefit from treatment with the immune checkpoint inhibitor drug pembrolizumab and PARP inhibitors, namely olaparib, rucaparib, or niraparib. Bone... 77 KB (8,858 words) - 14:12, 23 April 2024 |
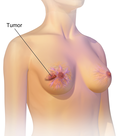 | for those whose tumors express PD-L1. And the similar immunotherapy pembrolizumab for those whose tumors have mutations in various DNA repair pathways... 133 KB (14,898 words) - 19:40, 5 May 2024 |
cancer, bladder cancer, head and neck cancer, and Hodgkin's lymphoma. Pembrolizumab (brand name Keytruda) is another PD-1 inhibitor that was approved by... 21 KB (2,093 words) - 18:53, 25 January 2024 |
relative with MS. Several other monoclonal antibodies like adalimumab, pembrolizumab, nivolumab, and infliximab have been reported to trigger MS as an adverse... 34 KB (3,827 words) - 06:58, 22 April 2024 |
 | pembrolizumab may have only minimal effects on the rate of death resulting from treatment or the rate at which the cancer advances. Pembrolizumab may... 137 KB (13,535 words) - 17:38, 29 April 2024 |
 | cancer with mismatch repair deficiency and microsatellite instability. Pembrolizumab is approved for advanced CRC tumours that are MMR deficient and have... 150 KB (16,285 words) - 20:19, 7 May 2024 |
accelerated cutaneous chronic wound healing (24 Mar), phase 3 trialed pembrolizumab addition against recurrent endometrial cancer (27 Mar), a phase 3 trialed... 324 KB (28,839 words) - 08:57, 22 April 2024 |
 | Pemetrexed is also recommended in combination with carboplatin and pembrolizumab for the first-line treatment of advanced non-small cell lung cancer... 17 KB (1,250 words) - 00:06, 31 March 2024 |
 | (e.g. sunitinib), checkpoint inhibitors (e.g. ipilimumab, nivolumab, pembrolizumab, atezolizumab) Contrast agent Gadolininum based contrast agent Other... 20 KB (2,303 words) - 11:41, 4 May 2024 |
 | Immunotherapy: mechanism of action for Durvalumab, Pembrolizumab, Ipilimumab, Atezolizumab . Adapted from: First line Immunotherapy for Non-Small Cell... 18 KB (1,668 words) - 03:20, 20 April 2024 |
chemoresistant MSI-high tumors, studies conducted on dostarlimab and pembrolizumab display promising results of the tumors reacting well to the therapies... 36 KB (3,089 words) - 05:24, 26 March 2024 |