 | Potassium azide is the inorganic compound having the formula KN3. It is a white, water-soluble salt. It is used as a reagent in the laboratory. It has... 7 KB (624 words) - 14:39, 20 April 2024 |
 | Lead(II) azide Pb(N3)2 is an inorganic compound. More so than other azides, it is explosive. It is used in detonators to initiate secondary explosives... 10 KB (880 words) - 16:17, 23 November 2023 |
 | Hydrazoic acid (redirect from Hydrogen azide) Hydrazoic acid, also known as hydrogen azide, azic acid or azoimide, is a compound with the chemical formula HN3. It is a colorless, volatile, and explosive... 12 KB (1,139 words) - 14:08, 30 December 2023 |
phenylmercury acetate picloram picolinafen pinoxaden piperophos potassium arsenite potassium azide potassium cyanate pretilachlor primisulfuron procyazine prodiamine... 14 KB (896 words) - 23:51, 29 December 2023 |
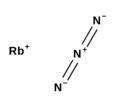 | H2O This formula is typically used to synthesize potassium azide from caustic potash. Rubidium azide has been investigated for possible use in alkali... 8 KB (704 words) - 23:28, 19 March 2024 |
KAsO2 Potassium azide – KN3 Potassium borate – K2B4O7·4H2O Potassium bromide – KBr Potassium bicarbonate – KHCO3 Potassium bifluoride – KHF2 Potassium bisulfite... 119 KB (8,726 words) - 04:34, 18 April 2024 |
piperalin polycarbamate polyoxins polyoxorim posaconazole potassium azide potassium polysulfide potassium thiocyanate pramiconazole probenazole prochloraz procymidone... 13 KB (891 words) - 16:16, 21 April 2024 |
378×10−14 Potassium acetate KC2H3O2 216 233 256 283 324 350 381 398 Potassium arsenate K3AsO4 19 Potassium azide KN3 41.4 46.2 50.8 55.8 61 106 Potassium benzoate... 90 KB (191 words) - 03:07, 1 November 2023 |
Pyrotechnic initiator (redirect from Zirconium-potassium perchlorate) relatively little harm to environment. It can be ignited by a laser diode. Lead azide (Pb(N3)2, or PbN6) is occasionally used in pyrotechnic initiators. Other... 12 KB (1,398 words) - 19:06, 21 October 2022 |
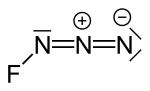 | Fluorine azide or triazadienyl fluoride is a yellow green gas composed of nitrogen and fluorine with formula FN3. Its properties resemble those of ClN3... 8 KB (724 words) - 12:54, 11 January 2024 |
KNO3 potassium nitrate 7757–79–1 KN3 potassium azide 20762–60–1 KNbO3 potassium niobate 12030–85–2 KOH potassium hydroxide 1310–58–3 KO2 potassium superoxide... 139 KB (120 words) - 19:28, 6 February 2024 |
However, the alkali metal azides NaN3 and KN3, featuring the linear N− 3 anion, are well-known, as are Sr(N3)2 and Ba(N3)2. Azides of the B-subgroup metals... 34 KB (4,297 words) - 09:04, 25 February 2024 |
US might contain 130 grams of sodium azide. Similar gas generators are used for fire suppression. Sodium azide decomposes exothermically to sodium and... 9 KB (890 words) - 17:41, 6 January 2024 |
dinitride Ethyl azide Explosive antimony Fluorine perchlorate Fulminic acid Halogen azides: Fluorine azide Chlorine azide Bromine azide Iodine azide Hexamethylene... 73 KB (8,326 words) - 08:56, 25 March 2024 |
 | 10–20% potassium chlorate is cheaper and more brisant than the fulminate alone. Silver fulminate is often confused with silver nitride, silver azide, or... 9 KB (948 words) - 15:35, 23 January 2024 |
 | decomposition of sodium azide. Potassium occurs in many minerals, such as sylvite (potassium chloride). Previously, potassium was generally made from... 214 KB (23,359 words) - 03:13, 22 April 2024 |
The azo and azide groups respectively, connected to organic/inorganic compounds (e.g. silver azide AgN3, lead azide Pb(N3)2, ammonium azide NH4N3) III... 3 KB (423 words) - 07:35, 27 January 2023 |
 | method includes two reaction steps with potentially hazardous azides. A reported azide-free Roche synthesis of tamiflu is summarised graphically below:... 20 KB (2,309 words) - 12:58, 25 October 2023 |
 | E2, while weaker bases that are still good nucleophiles (e.g., acetate, azide, cyanide, iodide) will give primarily SN2. Finally, weakly nucleophilic... 14 KB (1,843 words) - 07:43, 4 February 2024 |
Thiocarboxylic acids react with various nitrogen functional groups, such as organic azide, nitro, and isocyanate compounds, to give amides under mild conditions.... 5 KB (586 words) - 14:17, 27 December 2023 |










