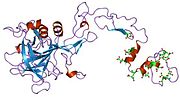
Recombinant factor VIIa (rfVIIa) is a form of blood factor VII that has been manufactured via recombinant technology. It is administered via an injection... 19 KB (1,756 words) - 15:27, 27 January 2024 |
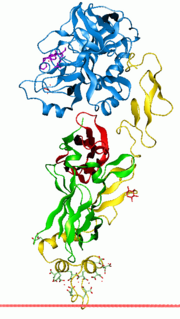 | factor VII), which in turn activates factor IX and factor X. Using genetic recombination a recombinant factor VIIa (eptacog alfa) (trade names include... 12 KB (1,376 words) - 17:01, 10 May 2024 |
plasma (FFP), vitamin K, prothrombin complex concentrates, or recombinant factor VIIa can be given to reverse the effect of warfarin. High doses of vitamin... 54 KB (5,859 words) - 15:59, 13 May 2024 |
 | trauma. For severe bleeding, for example from bleeding disorders, recombinant factor VIIa—a protein that assists blood clotting—may be appropriate. While... 59 KB (6,703 words) - 03:31, 12 April 2024 |
 | Anticoagulant (redirect from Anticoagulation factor) recombinant factor VIIa, and prothrombin complex concentrate (PCC) have been utilized with proven efficacy. Specifically with warfarin, four-factor PCC... 78 KB (8,726 words) - 15:18, 29 April 2024 |
 | Abdominal pregnancy (section Risk factors) kind of pregnancy, with others even using tranexamic acid and recombinant factor VIIa, which both minimize blood loss. Generally, unless the placenta... 26 KB (2,859 words) - 04:25, 5 May 2024 |
 | (only if bleeding is severe; risk of platelet alloimmunization) Recombinant factor VIIa, AryoSeven or NovoSeven FDA approved this drug for the treatment... 10 KB (1,027 words) - 15:47, 4 May 2024 |
trials to understand more about the potential benefits and risks. Recombinant factor VIIa (rFVIIa) is not, as of 2012, supported by the evidence for most... 20 KB (2,391 words) - 17:27, 30 March 2024 |
 | factor VIIa catalyzes the conversion of the inactive protease factor X into the active protease factor Xa. Together with factor VIIa, tissue factor forms... 15 KB (1,833 words) - 16:50, 10 May 2024 |
Acquired haemophilia (redirect from Anti-factor VIII autoimmunization) Comparative thrombotic event incidence after infusion of recombinant factor VIIa versus factor VIII inhibitor bypass activity. J Thromb Haemost 2004; 2:1700-1708... 10 KB (1,468 words) - 18:35, 1 October 2022 |
hemophilia A and rare coagulation disorders. Patterns of response to recombinant factor VIIa. J Thromb Haemost. 2004;2:102-10. Karger R, Egbring R, Weippert-Kretschmer... 19 KB (2,428 words) - 14:04, 5 December 2022 |
 | Coagulation (redirect from Coagulation factor) fresh frozen plasma are commonly used coagulation factor products. Recombinant activated human factor VII is increasingly popular in the treatment of major... 61 KB (5,960 words) - 10:29, 11 May 2024 |
 | 1980s. Lusher later conducted the first studies on the use of recombinant factor VIIa in the treatment of bleeding episodes in patients with hemophilia... 14 KB (1,906 words) - 06:26, 4 June 2023 |
 | between intensive and less intensive blood pressure control. Giving Factor VIIa within 4 hours limits the bleeding and formation of a hematoma. However... 44 KB (4,483 words) - 13:03, 4 May 2024 |
 | Amniotic fluid embolism (section Risk factors) promptly to prevent further complications. In the case of DIC, recombinant activated factor VIIa is quick way to address this issue. Serine proteinase inhibitor... 18 KB (1,947 words) - 11:31, 26 March 2024 |
investigator of a multinational multicenter testing program evaluating recombinant factor VIIa for this indication. Mayer predominantly uses invasive brain multimodality... 17 KB (1,770 words) - 02:46, 22 November 2023 |
 | Factor II (thrombin) (IIa), and also the activated form of Factor VII (VIIa) from the tissue factor pathway (formerly known as the extrinsic pathway). The... 45 KB (5,331 words) - 23:48, 9 March 2024 |
2008, the company sold its MAXY-VII candidate drug, a recombinant form of clotting factor VIIa, to Bayer HealthCare. 2009 saw the formation of a joint... 9 KB (880 words) - 02:18, 3 February 2023 |
slow, tight-binding inhibitor of factor Xa with a Ki value of 0.3–0.6 nM but it also inhibits trypsin. Recombinant Antistasin can be produced by genetically... 41 KB (4,917 words) - 22:24, 10 May 2024 |
development and commercialization of three drug candidates, recombinant blood clotting factors VIIa, VIII and IX, the latter of which is in clinical trials... 10 KB (885 words) - 10:13, 28 February 2024 |
– factor va MeSH D23.119.325.300 – factor viia MeSH D23.119.350.300 – factor viiia MeSH D23.119.375.310 – factor ixa MeSH D23.119.400.315 – factor xa... 86 KB (8,039 words) - 16:46, 9 February 2024 |