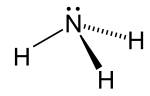
| Thermochemistry is the study of the heat energy which is associated with chemical reactions and/or phase changes such as melting and boiling. A reaction... 7 KB (788 words) - 00:11, 12 February 2024 |
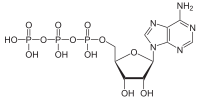 | Adenosine triphosphate (redirect from ATP thermochemistry) Adenosine triphosphate (ATP) is a nucleotide that provides energy to drive and support many processes in living cells, such as muscle contraction, nerve... 46 KB (5,062 words) - 17:08, 19 April 2024 |
 | laws, Kirchhoff's law of thermal radiation, and Kirchhoff's law of thermochemistry. The Bunsen–Kirchhoff Award for spectroscopy is named after Kirchhoff... 19 KB (1,729 words) - 16:55, 1 April 2024 |
Outline of chemistry (section Thermochemistry) solid–gas interfaces, solid–vacuum interfaces, and liquid-gas interfaces. Thermochemistry –The branch of chemistry that studies the relation between chemical... 33 KB (4,048 words) - 23:47, 7 February 2024 |
dynamics Kirchhoff's three laws of spectroscopy Kirchhoff's law of thermochemistry Kirchhoff's theorem about the number of spanning trees in a graph Kerckhoffs's... 661 bytes (91 words) - 13:24, 11 September 2023 |
which the standard Gibbs free energy of formation can be calculated. Thermochemistry Calorimetry M. W. Chase, NIST – JANAF Thermochemical Tables, 4th Edition... 9 KB (201 words) - 07:30, 9 April 2024 |
 | (16 February 1826 – 13 February 1909) was a Danish chemist noted in thermochemistry for the Thomsen–Berthelot principle. Thomsen was born in Copenhagen... 7 KB (604 words) - 20:45, 19 December 2023 |
 | chemist and doctor who formulated Hess's law, an early principle of thermochemistry. Hess was born on 7 August 1802 in Geneva, Switzerland. His father... 8 KB (789 words) - 20:13, 10 May 2023 |
Syngas, or synthesis gas, is a mixture of hydrogen and carbon monoxide, in various ratios. The gas often contains some carbon dioxide and methane. It is... 15 KB (1,414 words) - 11:29, 29 March 2024 |
exchange. Intended for main group thermochemistry and non-covalent interactions, transition metal thermochemistry and organometallics. It is usually... 26 KB (2,521 words) - 09:54, 21 March 2024 |
 | 1.377 nβ = 1.501 nγ = 1.583 Structure Crystal structure Monoclinic Thermochemistry Heat capacity (C) 87.6 J/mol K Std molar entropy (S⦵298) 101.7 J/mol... 52 KB (4,960 words) - 10:11, 29 April 2024 |
Physical organic chemistry (section Thermochemistry) Benson who spent a career developing the concept.[page needed] The thermochemistry of reactive intermediates—carbocations, carbanions, and radicals—is... 50 KB (5,580 words) - 08:24, 7 February 2024 |
 | Exothermic reaction (category Thermochemistry) In thermochemistry, an exothermic reaction is a "reaction for which the overall standard enthalpy change ΔH⚬ is negative." Exothermic reactions usually... 5 KB (552 words) - 07:35, 19 March 2024 |
Principle of maximum work (category Thermochemistry) form in 1875 by French chemist Marcellin Berthelot, in the field of thermochemistry, and then in 1876 by American mathematical physicist Willard Gibbs... 7 KB (907 words) - 23:48, 23 September 2022 |
 | g/cm3 Thermal conductivity 0.2 W/(m·K) Refractive index (nD) 1.63 Thermochemistry Heat capacity (C) 0.92 kJ/(kg·K) Except where otherwise noted, data... 45 KB (4,595 words) - 11:11, 29 April 2024 |
substance. For example, the Gibbs free energy of a compound in the area of thermochemistry is often quantified in units of kilojoules per mole (symbol: kJ·mol−1... 4 KB (486 words) - 19:24, 30 August 2023 |
 | log P −3.76 Structure Crystal structure Monoclinic Space group P21 Thermochemistry Std enthalpy of formation (ΔfH⦵298) −2,226.1 kJ/mol (−532.1 kcal/mol)... 64 KB (7,038 words) - 09:26, 21 April 2024 |
(20 °C) Structure Molecular shape Tetrahedral Dipole moment 1.15 D Thermochemistry Heat capacity (C) 114.25 J/(mol·K) Std molar entropy (S⦵298) 202.9... 59 KB (5,618 words) - 13:17, 26 April 2024 |
 | surface. A particularly important objective, called computational thermochemistry, is to calculate thermochemical quantities such as the enthalpy of... 77 KB (8,359 words) - 02:44, 27 March 2024 |