Titanium dioxide
 | |
 | |
| Names | |
|---|---|
| IUPAC names Titanium dioxide Titanium(IV) oxide | |
| Other names | |
| Identifiers | |
3D model (JSmol) | |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.033.327 |
| E number | E171 (colours) |
| KEGG | |
PubChem CID | |
| RTECS number |
|
| UNII | |
CompTox Dashboard (EPA) | |
| |
| |
| Properties | |
| TiO 2 | |
| Molar mass | 79.866 g/mol |
| Appearance | White solid |
| Odor | Odorless |
| Density |
|
| Melting point | 1,843 °C (3,349 °F; 2,116 K) |
| Boiling point | 2,972 °C (5,382 °F; 3,245 K) |
| Insoluble | |
| Band gap | 3.21 eV (anatase)[1] 3.15 eV (rutile)[1] |
| +5.9·10−6 cm3/mol | |
Refractive index (nD) |
|
| Thermochemistry | |
Std molar entropy (S⦵298) | 50 J·mol−1·K−1[2] |
Std enthalpy of formation (ΔfH⦵298) | −945 kJ·mol−1[2] |
| Hazards | |
| NFPA 704 (fire diamond) | |
| Flash point | not flammable |
| NIOSH (US health exposure limits): | |
PEL (Permissible) | TWA 15 mg/m3[3] |
REL (Recommended) | Ca[3] |
IDLH (Immediate danger) | Ca [5000 mg/m3][3] |
| Safety data sheet (SDS) | ICSC 0338 |
| Related compounds | |
Other cations | Zirconium dioxide Hafnium dioxide |
| Titanium(II) oxide Titanium(III) oxide Titanium(III,IV) oxide | |
Related compounds | Titanic acid |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
Titanium dioxide, also known as titanium(IV) oxide or titania /taɪˈteɪniə/, is the inorganic compound derived from titanium with the chemical formula TiO
2. When used as a pigment, it is called titanium white, Pigment White 6 (PW6), or CI 77891.[4] It is a white solid that is insoluble in water, although mineral forms can appear black. As a pigment, it has a wide range of applications, including paint, sunscreen, and food coloring. When used as a food coloring, it has E number E171. World production in 2014 exceeded 9 million tonnes.[5][6][7] It has been estimated that titanium dioxide is used in two-thirds of all pigments, and pigments based on the oxide have been valued at a price of $13.2 billion.[8]
Structure
[edit]
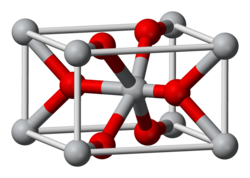
In all three of its main dioxides, titanium exhibits octahedral geometry, being bonded to six oxide anions. The oxides in turn are bonded to three Ti centers. The overall crystal structures of rutile and anatase are tetragonal in symmetry whereas brookite is orthorhombic. The oxygen substructures are all slight distortions of close packing: in rutile, the oxide anions are arranged in distorted hexagonal close-packing, whereas they are close to cubic close-packing in anatase and to "double hexagonal close-packing" for brookite. The rutile structure is widespread for other metal dioxides and difluorides, e.g. RuO2 and ZnF2.
Molten titanium dioxide has a local structure in which each Ti is coordinated to, on average, about 5 oxygen atoms.[9] This is distinct from the crystalline forms in which Ti coordinates to 6 oxygen atoms.
Synthetic and geologic occurrence
[edit]Synthetic TiO2 is mainly produced from the mineral ilmenite. Rutile, and anatase, naturally occurring TiO2, occur widely also, e.g. rutile as a 'heavy mineral' in beach sand. Leucoxene, fine-grained anatase formed by natural alteration of ilmenite, is yet another ore. Star sapphires and rubies get their asterism from oriented inclusions of rutile needles.[10]
Mineralogy and uncommon polymorphs
[edit]Titanium dioxide occurs in nature as the minerals rutile and anatase. Additionally two high-pressure forms are known minerals: a monoclinic baddeleyite-like form known as akaogiite, and the other has a slight monoclinic distortion of the orthorhombic α-PbO2 structure and is known as riesite. Both of which can be found at the Ries crater in Bavaria.[11][12][13] It is mainly sourced from ilmenite, which is the most widespread titanium dioxide-bearing ore around the world. Rutile is the next most abundant and contains around 98% titanium dioxide in the ore. The metastable anatase and brookite phases convert irreversibly to the equilibrium rutile phase upon heating above temperatures in the range 600–800 °C (1,110–1,470 °F).[14]
Titanium dioxide has twelve known polymorphs – in addition to rutile, anatase, brookite, akaogiite and riesite, three metastable phases can be produced synthetically (monoclinic, tetragonal, and orthorhombic ramsdellite-like), and four high-pressure forms (α-PbO2-like, cotunnite-like, orthorhombic OI, and cubic phases) also exist:
| Form | Crystal system | Synthesis |
|---|---|---|
| Rutile | Tetragonal | |
| Anatase | Tetragonal | |
| Brookite | Orthorhombic | |
| TiO2(B)[15] | Monoclinic | Hydrolysis of K2Ti4O9 followed by heating |
| TiO2(H), hollandite-like form[16] | Tetragonal | Oxidation of the related potassium titanate bronze, K0.25TiO2 |
| TiO2(R), ramsdellite-like form[17] | Orthorhombic | Oxidation of the related lithium titanate bronze Li0.5TiO2 |
| TiO2(II)-(α-PbO2-like form)[18] | Orthorhombic | |
| Akaogiite (baddeleyite-like form, 7 coordinated Ti)[19] | Monoclinic | |
| TiO2 -OI[20] | Orthorhombic | |
| Cubic form[21] | Cubic | P > 40 GPa, T > 1600 °C |
| TiO2 -OII, cotunnite(PbCl2)-like[22] | Orthorhombic | P > 40 GPa, T > 700 °C |
The cotunnite-type phase was claimed to be the hardest known oxide with the Vickers hardness of 38 GPa and the bulk modulus of 431 GPa (i.e. close to diamond's value of 446 GPa) at atmospheric pressure.[22] However, later studies came to different conclusions with much lower values for both the hardness (7–20 GPa, which makes it softer than common oxides like corundum Al2O3 and rutile TiO2)[23] and bulk modulus (~300 GPa).[24][25]
Titanium dioxide (B) is found as a mineral in magmatic rocks and hydrothermal veins, as well as weathering rims on perovskite. TiO2 also forms lamellae in other minerals.[26]
Production
[edit]

The largest TiO
2 pigment processors are Chemours, Venator, Kronos, and Tronox.[27][28] Major paint and coating company end users for pigment grade titanium dioxide include Akzo Nobel, PPG Industries, Sherwin Williams, BASF, Kansai Paints and Valspar.[29] Global TiO
2 pigment demand for 2010 was 5.3 Mt with annual growth expected to be about 3–4%.[30]
The production method depends on the feedstock. In addition to ores, other feedstocks include upgraded slag. Both the chloride process and the sulfate process (both described below) produce titanium dioxide pigment in the rutile crystal form, but the sulfate process can be adjusted to produce the anatase form. Anatase, being softer, is used in fiber and paper applications. The sulfate process is run as a batch process; the chloride process is run as a continuous process.[31]
Chloride process
[edit]In chloride process, the ore is treated with chlorine and carbon to give titanium tetrachloride, a volatile liquid that is further purified by distillation. The TiCl4 is treated with oxygen to regenerate chlorine and produce the titanium dioxide.
Sulfate process
[edit]In the sulfate process, ilmenite is treated with sulfuric acid to extract iron(II) sulfate pentahydrate. This process requires concentrated ilmenite (45–60% TiO2) or pretreated feedstocks as a suitable source of titanium.[32] The resulting synthetic rutile is further processed according to the specifications of the end user, i.e. pigment grade or otherwise.[33]
Examples of plants using the sulfate process are the Sorel-Tracy plant of QIT-Fer et Titane and the Eramet Titanium & Iron smelter in Tyssedal Norway.[34]
Becher process
[edit]The Becher process is another method for the production of synthetic rutile from ilmenite. It first oxidizes the ilmenite as a means to separate the iron component.
Specialized methods
[edit]For specialty applications, TiO2 films are prepared by various specialized chemistries.[35] Sol-gel routes involve the hydrolysis of titanium alkoxides such as titanium ethoxide:
- Ti(OEt)4 + 2 H2O → TiO2 + 4 EtOH
A related approach that also relies on molecular precursors involves chemical vapor deposition. In this method, the alkoxide is volatilized and then decomposed on contact with a hot surface:
- Ti(OEt)4 → TiO2 + 2 Et2O
Applications
[edit]Pigment
[edit]First mass-produced in 1916,[36] titanium dioxide is the most widely used white pigment because of its brightness and very high refractive index, in which it is surpassed only by a few other materials (see list of indices of refraction). Titanium dioxide crystal size is ideally around 220 nm (measured by electron microscope) to optimize the maximum reflection of visible light. However, abnormal grain growth is often observed in titanium dioxide, particularly in its rutile phase.[37] The occurrence of abnormal grain growth brings about a deviation of a small number of crystallites from the mean crystal size and modifies the physical behaviour of TiO2. The optical properties of the finished pigment are highly sensitive to purity. As little as a few parts per million (ppm) of certain metals (Cr, V, Cu, Fe, Nb) can disturb the crystal lattice so much that the effect can be detected in quality control.[38][full citation needed] Approximately 4.6 million tons of pigmentary TiO2 are used annually worldwide, and this number is expected to increase as use continues to rise.[39]
TiO2 is also an effective opacifier in powder form, where it is employed as a pigment to provide whiteness and opacity to products such as paints, coatings, plastics, papers, inks, foods, supplements, medicines (i.e. pills and tablets), and most toothpastes; in 2019 it was present in two-thirds of toothpastes on the French market.[40] In paint, it is often referred to offhandedly as "brilliant white", "the perfect white", "the whitest white", or other similar terms. Opacity is improved by optimal sizing of the titanium dioxide particles.
Additive for food
[edit]Often used as color in food,[41] it is commonly found in ice creams, chocolates, all types of candy, creamers, desserts, marshmallows, chewing gum, pastries, spreads, dressings, cakes, some cheeses, and many other foods.[42] It is permitted in many countries, but was banned for use in food by the European Union in 2022. While permitted in the United States, Mars removed it from their Skittles confectionery in 2025, although a class-action lawsuit against the use of titanium dioxide in Skittles had been dismissed in 2022.[43]
Thin films
[edit]When deposited as a thin film, its refractive index and colour make it an excellent reflective optical coating for dielectric mirrors; it is also used in generating decorative thin films such as found in "mystic fire topaz".[citation needed]
Some grades of modified titanium based pigments as used in sparkly paints, plastics, finishes and cosmetics – these are man-made pigments whose particles have two or more layers of various oxides – often titanium dioxide, iron oxide or alumina – in order to have glittering, iridescent and or pearlescent effects similar to crushed mica or guanine-based products. In addition to these effects a limited colour change is possible in certain formulations depending on how and at which angle the finished product is illuminated and the thickness of the oxide layer in the pigment particle; one or more colours appear by reflection while the other tones appear due to interference of the transparent titanium dioxide layers.[44] In some products, the layer of titanium dioxide is grown in conjunction with iron oxide by calcination of titanium salts (sulfates, chlorates) around 800 °C[45] One example of a pearlescent pigment is Iriodin, based on mica coated with titanium dioxide or iron (III) oxide.[46]
The iridescent effect in these titanium oxide particles is unlike the opaque effect obtained with usual ground titanium oxide pigment obtained by mining, in which case only a certain diameter of the particle is considered and the effect is due only to scattering.
Sunscreen and UV blocking pigments
[edit]In cosmetic and skin care products, titanium dioxide is used as a pigment, sunscreen and a thickener. As a sunscreen, ultrafine TiO2 is used, which is notable in that combined with ultrafine zinc oxide, it is considered to be an effective sunscreen that lowers the incidence of sun burns and minimizes the premature photoaging, photocarcinogenesis and immunosuppression associated with long term excess sun exposure.[47] Sometimes these UV blockers are combined with iron oxide pigments in sunscreen to increase visible light protection.[48]
Titanium dioxide and zinc oxide are generally considered to be less harmful to coral reefs than sunscreens that include chemicals such as oxybenzone, octocrylene and octinoxate.[49]
Nanosized titanium dioxide is found in the majority of physical sunscreens because of its strong UV light absorbing capabilities and its resistance to discolouration under ultraviolet light. This advantage enhances its stability and ability to protect the skin from ultraviolet light. Nano-scaled (particle size of 20–40 nm)[50] titanium dioxide particles are primarily used in sunscreen lotion because they scatter visible light much less than titanium dioxide pigments, and can give UV protection.[39] Sunscreens designed for infants or people with sensitive skin are often based on titanium dioxide and/or zinc oxide, as these mineral UV blockers are believed to cause less skin irritation than other UV absorbing chemicals. Nano-TiO2, which blocks both UV-A and UV-B radiation, is used in sunscreens and other cosmetic products.
The EU Scientific Committee on Consumer Safety considered nano sized titanium dioxide to be safe for skin applications, in concentrations of up to 25 percent based on animal testing.[51] The risk assessment of different titanium dioxide nanomaterials in sunscreen is currently evolving since nano-sized TiO2 is different from the well-known micronized form.[52] The rutile form is generally used in cosmetic and sunscreen products due to it not possessing any observed ability to damage the skin under normal conditions[53] and having a higher UV absorption.[54] In 2016 Scientific Committee on Consumer Safety (SCCS) tests concluded that the use of nano titanium dioxide (95–100% rutile, ≦5% anatase) as a UV filter can be considered to not pose any risk of adverse effects in humans post-application on healthy skin,[55] except in the case the application method would lead to substantial risk of inhalation (ie; powder or spray formulations). This safety opinion applied to nano TiO2 in concentrations of up to 25%.[56]
Initial studies indicated that nano-TiO2 particles could penetrate the skin, causing concern over its use. These studies were later refuted, when it was discovered that the testing methodology couldn't differentiate between penetrated particles and particles simply trapped in hair follicles and that having a diseased or physically damaged dermis could be the true cause of insufficient barrier protection.[52]
SCCS research found that when nanoparticles had certain photostable coatings (e.g., alumina, silica, cetyl phosphate, triethoxycaprylylsilane, manganese dioxide), the photocatalytic activity was attenuated and no notable skin penetration was observed; the sunscreen in this research was applied at amounts of 10 mg/cm2 for exposure periods of 24 hours.[56] Coating TiO2 with alumina, silica, zircon or various polymers can minimize avobenzone degradation[57] and enhance UV absorption by adding an additional light diffraction mechanism.[54]
TiO
2 is used extensively in plastics and other applications as a white pigment or an opacifier and for its UV resistant properties where the powder disperses light – unlike organic UV absorbers – and reduces UV damage, due mostly to the particle's high refractive index.[58]
Other uses of titanium dioxide
[edit]In ceramic glazes, titanium dioxide acts as an opacifier and seeds crystal formation.
It is used as a tattoo pigment and in styptic pencils. Titanium dioxide is produced in varying particle sizes which are both oil and water dispersible, and in certain grades for the cosmetic industry. It is also a common ingredient in toothpaste.
The exterior of the Saturn V rocket was painted with titanium dioxide; this later allowed astronomers to determine that J002E3 was likely the S-IVB stage from Apollo 12 and not an asteroid.[59]
Titanium dioxide is an n-type semiconductor and is used in dye-sensitized solar cells.[60] It is also used in other electronics components such as electrodes in batteries.[61]
Research
[edit]Patenting activities
[edit]

Between 2002 and 2022, there were 459 patent families that describe the production of titanium dioxide from ilmenite. The majority of these patents describe pre-treatment processes, such as using smelting and magnetic separation to increase titanium concentration in low-grade ores, leading to titanium concentrates or slags. Other patents describe processes to obtain titanium dioxide, either by a direct hydrometallurgical process or through the main industrial production processes, the sulfate process and the chloride process.[62] The sulfate process represents 40% of the world’s titanium dioxide production and is protected in 23% of patent families. The chloride process is only mentioned in 8% of patent families, although it provides 60% of the worldwide industrial production of titanium dioxide.[62]
Key contributors to patents on the production of titanium dioxide are companies from China, Australia and the United States, reflecting the major contribution of these countries to industrial production. Chinese companies Pangang and Lomon Billions Groups hold major patent portfolios.[62]
Photocatalyst
[edit]Nanosized titanium dioxide, particularly in the anatase form, exhibits photocatalytic activity under ultraviolet (UV) irradiation. This photoactivity is reportedly most pronounced at the {001} planes of anatase,[63][64] although the {101} planes are thermodynamically more stable and thus more prominent in most synthesised and natural anatase,[65] as evident by the often observed tetragonal dipyramidal growth habit. Interfaces between rutile and anatase are further considered to improve photocatalytic activity by facilitating charge carrier separation and as a result, biphasic titanium dioxide is often considered to possess enhanced functionality as a photocatalyst.[66] It has been reported that titanium dioxide, when doped with nitrogen ions or doped with metal oxide like tungsten trioxide, exhibits excitation also under visible light.[67] The strong oxidative potential of the positive holes oxidizes water to create hydroxyl radicals. It can also oxidize oxygen or organic materials directly. Hence, in addition to its use as a pigment, titanium dioxide can be added to paints, cements, windows, tiles, or other products for its sterilizing, deodorizing, and anti-fouling properties, and is used as a hydrolysis catalyst. It is also used in dye-sensitized solar cells, which are a type of chemical solar cell (also known as a Graetzel cell).
The photocatalytic properties of nanosized titanium dioxide were discovered by Akira Fujishima in 1967[68] and published in 1972.[69] The process on the surface of the titanium dioxide was called the Honda-Fujishima effect.[68] In thin film and nanoparticle form, titanium dioxide has the potential for use in energy production: As a photocatalyst, it can break water into hydrogen and oxygen. With the hydrogen collected, it could be used as a fuel. The efficiency of this process can be greatly improved by doping the oxide with carbon.[70] Further efficiency and durability has been obtained by introducing disorder to the lattice structure of the surface layer of titanium dioxide nanocrystals, permitting infrared absorption.[71] Visible-light-active nanosized anatase and rutile has been developed for photocatalytic applications.[72][73]
In 1995 Fujishima and his group discovered the superhydrophilicity phenomenon for titanium dioxide coated glass exposed to sun light.[68] This resulted in the development of self-cleaning glass and anti-fogging coatings.
Nanosized TiO2 incorporated into outdoor building materials, such as paving stones in noxer blocks[74] or paints, could reduce concentrations of airborne pollutants such as volatile organic compounds and nitrogen oxides.[75] A TiO2-containing cement has been produced.[76]
Using TiO2 as a photocatalyst, attempts have been made to mineralize pollutants (to convert into CO2 and H2O) in waste water.[77][78][79] The photocatalytic destruction of organic matter could also be exploited in coatings with antimicrobial applications.[80]
Hydroxyl radical formation
[edit]Although nanosized anatase TiO2 does not absorb visible light, it does strongly absorb ultraviolet (UV) radiation (hv), leading to the formation of hydroxyl radicals.[81] This occurs when photo-induced valence bond holes (h+vb) are trapped at the surface of TiO2 leading to the formation of trapped holes (h+tr) that cannot oxidize water.[82]
- TiO2 + hv → e− + h+vb
- h+vb → h+tr
- O2 + e− → O2•−
- O2•− + O2•−+ 2 H+ → H2O2 + O2
- O2•− + h+vb → O2
- O2•− + h+tr → O2
- OH− + h+vb → HO•
- e− + h+tr → recombination
- Note: Wavelength (λ)= 387 nm[82] This reaction has been found to mineralize and decompose undesirable compounds in the environment, specifically the air and in wastewater.[82]

Nanotubes
[edit]Anatase can be converted into non-carbon nanotubes and nanowires.[83] Hollow TiO2 nanofibers can be also prepared by coating carbon nanofibers by first applying titanium butoxide.[84]
- Titanium oxide nanotubes, SEM image
- Nanotubes of titanium dioxide (TiO2-Nt) obtained by electrochemical synthesis. The SEM image shows an array of vertical self-ordered TiO2-Nt with closed bottom ends of tubes.
Solubility
[edit]Titanium dioxide is insoluble in water, organic solvents, and inorganic acids. It is slightly soluble in alkali, soluble in saturated potassium acid carbonate, and can be completely dissolved in strong sulfuric acid and hydrofluoric acid after boiling for a long time.[85]
Health and safety
[edit]Widely-occurring minerals and even gemstones are composed of TiO2. All natural titanium, comprising more than 0.5% of the Earth's crust, exists as oxides.[86]
Food additive
[edit]As of 2024, titanium dioxide is considered safe by the US FDA as a color ingredient for oral human consumption as long as it is 1% or less of the total food composition.[87] A 2021 ban by the EU EFSA has been criticized as based on errors regarding the safety of titanium dioxide (E171) particles as a food additive,[88] and according to a 2022 review, existing evidence does not support a direct DNA damaging mechanism for titanium dioxide.[89]
Government policies
[edit]TiO2 whitener in food was banned in France from 2020, due to uncertainty about safe quantities for human consumption.[90]
In 2021, the European Food Safety Authority (EFSA) ruled that as a consequence of new understandings of nanoparticles, titanium dioxide could "no longer be considered safe as a food additive", and the EU health commissioner announced plans to ban its use across the EU, with discussions beginning in June 2021. EFSA concluded that genotoxicity—which could lead to carcinogenic effects—could not be ruled out, and that a "safe level for daily intake of the food additive could not be established".[91] In 2022, the UK Food Standards Agency and Food Standards Scotland announced their disagreement with the EFSA ruling, and did not follow the EU in banning titanium dioxide as a food additive.[92] Health Canada similarly reviewed the available evidence in 2022 and decided not to change their position on titanium dioxide as a food additive.[93]
The European Union removed the authorization to use titanium dioxide (E 171) in foods, effective 7 February 2022, with a six months grace period.[94]
As of May 2023, following the European Union 2022 ban, the U.S. states California and New York were considering banning the use of titanium dioxide in foods.[95]
As of 2024, the Food and Drug Administration (FDA) in the United States permits titanium dioxide as a food additive.[87] It may be used to increase whiteness and opacity in dairy products (some cheeses, ice cream, and yogurt), candies, frostings, fillings, and many other foods. The FDA regulates the labeling of products containing titanium dioxide, allowing the product's ingredients list to identify titanium dioxide either as "color added" or "artificial colors" or "titanium dioxide;" it does not require that titanium dioxide be explicitly named.[87] In 2023, the Consumer Healthcare Products Association, a manufacturer's trade group, defended the substance as safe at certain limits while allowing that additional studies could provide further insight, saying an immediate ban would be a "knee-jerk" reaction.[96]
Industry response
[edit]Dunkin' Donuts dropped titanium dioxide from their merchandise in 2015 after public pressure.[97]
Research as an ingestible nanomaterial
[edit]Size distribution analyses showed that batches of food-grade TiO₂, which is produced with a target particle size in the 200–300 nm range for optimal pigmentation qualities, include a nanoparticle-sized fraction as inevitable byproduct of the manufacturing processes.[98]
Inhalation
[edit]Titanium dioxide dust, when inhaled, has been classified by the International Agency for Research on Cancer (IARC) as an IARC Group 2B carcinogen, meaning it is possibly carcinogenic to humans.[99][100] The US National Institute for Occupational Safety and Health recommends two separate exposure limits. NIOSH recommends that fine TiO
2 particles be set at an exposure limit of 2.4 mg/m3, while ultrafine TiO
2 be set at an exposure limit of 0.3 mg/m3, as time-weighted average concentrations up to 10 hours a day for a 40-hour work week.[101]
Although no evidence points to acute toxicity, recurring concerns have been expressed about nanophase forms of these materials. Studies of workers with high exposure to TiO2 particles indicate that even at high exposure there is no adverse effect to human health.[86]
Environmental waste introduction
[edit]Titanium dioxide (TiO₂) is mostly introduced into the environment as nanoparticles via wastewater treatment plants.[102] Cosmetic pigments including titanium dioxide enter the wastewater when the product is washed off into sinks after cosmetic use. Once in the sewage treatment plants, pigments separate into sewage sludge which can then be released into the soil when injected into the soil or distributed on its surface. 99% of these nanoparticles wind up on land rather than in aquatic environments due to their retention in sewage sludge.[102] In the environment, titanium dioxide nanoparticles have low to negligible solubility and have been shown to be stable once particle aggregates are formed in soil and water surroundings.[102] In the process of dissolution, water-soluble ions typically dissociate from the nanoparticle into solution when thermodynamically unstable. TiO2 dissolution increases when there are higher levels of dissolved organic matter and clay in the soil. However, aggregation is promoted by pH at the isoelectric point of TiO2 (pH= 5.8) which renders it neutral and solution ion concentrations above 4.5 mM.[103][104]
See also
[edit]- Delustrant
- Dye-sensitized solar cell
- List of inorganic pigments
- Noxer blocks, TiO2-coated pavers that remove NOx pollutants from the air
- Suboxide
- Surface properties of transition metal oxides
- Titanium dioxide nanoparticle
Sources
[edit]![]() This article incorporates text from a free content work. Licensed under CC-BY. Text taken from Production of titanium and titanium dioxide from ilmenite and related applications, WIPO.
This article incorporates text from a free content work. Licensed under CC-BY. Text taken from Production of titanium and titanium dioxide from ilmenite and related applications, WIPO.
References
[edit]- ^ a b Zanatta A (May 2024). "Temperature-dependent optical bandgap of TiO2 under the Anatase and Rutile phases". Results Phys. 60: 107653–5pp. doi:10.1016/j.rinp.2024.107653.
- ^ a b Zumdahl, Steven S. (2009). Chemical Principles 6th Ed. Houghton Mifflin Company. p. A23. ISBN 978-0-618-94690-7.
- ^ a b c NIOSH Pocket Guide to Chemical Hazards. "#0617". National Institute for Occupational Safety and Health (NIOSH).
- ^ Völz, Hans G., et al. (2006). "Pigments, Inorganic". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a20_243.pub2. ISBN 978-3-527-30673-2.
- ^ "Titanium" in 2014 Minerals Yearbook. USGS
- ^ "Mineral Commodity Summaries, 2015" (PDF). U.S. Geological Survey. U.S. Geological Survey 2015.
- ^ "Mineral Commodity Summaries, January 2016" (PDF). U.S. Geological Survey. U.S. Geological Survey 2016.
- ^ Schonbrun Z. "The Quest for the Next Billion-Dollar Color". Bloomberg.com. Retrieved 24 April 2018.
- ^ Alderman OL, Skinner LB, Benmore CJ, Tamalonis A, Weber JK (2014). "Structure of molten titanium dioxide". Physical Review B. 90 (9): 094204. Bibcode:2014PhRvB..90i4204A. doi:10.1103/PhysRevB.90.094204. ISSN 1098-0121.
- ^ Emsley J (2001). Nature's Building Blocks: An A–Z Guide to the Elements. Oxford: Oxford University Press. pp. 451–53. ISBN 978-0-19-850341-5.
- ^ El, Goresy, Chen, M, Dubrovinsky, L, Gillet, P, Graup, G (2001). "An ultradense polymorph of rutile with seven-coordinated titanium from the Ries crater". Science. 293 (5534): 1467–70. Bibcode:2001Sci...293.1467E. doi:10.1126/science.1062342. PMID 11520981. S2CID 24349901.
- ^ El Goresy, Ahmed, Chen M, Gillet P, Dubrovinsky L, Graup G, Ahuja R (2001). "A natural shock-induced dense polymorph of rutile with α-PbO2 structure in the suevite from the Ries crater in Germany". Earth and Planetary Science Letters. 192 (4): 485. Bibcode:2001E&PSL.192..485E. doi:10.1016/S0012-821X(01)00480-0.
- ^ Akaogiite. mindat.org
- ^ Hanaor DA, Sorrell CC (February 2011). "Review of the anatase to rutile phase transformation". Journal of Materials Science. 46 (4): 855–874. Bibcode:2011JMatS..46..855H. doi:10.1007/s10853-010-5113-0. S2CID 97190202.
- ^ Marchand R., Brohan L., Tournoux M. (1980). "A new form of titanium dioxide and the potassium octatitanate K2Ti8O17". Materials Research Bulletin. 15 (8): 1129–1133. doi:10.1016/0025-5408(80)90076-8.
- ^ Latroche, M, Brohan, L, Marchand, R, Tournoux (1989). "New hollandite oxides: TiO2(H) and K0.06TiO2". Journal of Solid State Chemistry. 81 (1): 78–82. Bibcode:1989JSSCh..81...78L. doi:10.1016/0022-4596(89)90204-1.
- ^ Akimoto J, Gotoh Y, Oosawa Y, Nonose N, Kumagai T, Aoki K, Takei H (1994). "Topotactic Oxidation of Ramsdellite-Type Li0.5TiO2, a New Polymorph of Titanium Dioxide: TiO2(R)". Journal of Solid State Chemistry. 113 (1): 27–36. Bibcode:1994JSSCh.113...27A. doi:10.1006/jssc.1994.1337.
- ^ Simons PY, Dachille F (1967). "The structure of TiO2II, a high-pressure phase of TiO2". Acta Crystallographica. 23 (2): 334–336. Bibcode:1967AcCry..23..334S. doi:10.1107/S0365110X67002713.
- ^ Sato H, Endo S, Sugiyama M, Kikegawa T, Shimomura O, Kusaba K (1991). "Baddeleyite-Type High-Pressure Phase of TiO2". Science. 251 (4995): 786–788. Bibcode:1991Sci...251..786S. doi:10.1126/science.251.4995.786. PMID 17775458. S2CID 28241170.
- ^ Dubrovinskaia N. A., Dubrovinsky L. S., Ahuja R., Prokopenko V. B., Dmitriev V., Weber H.-P., Osorio-Guillen J. M., Johansson B. (2001). "Experimental and Theoretical Identification of a New High-Pressure TiO2 Polymorph". Phys. Rev. Lett. 87 (27 Pt 1): 275501. Bibcode:2001PhRvL..87A5501D. doi:10.1103/PhysRevLett.87.275501. PMID 11800890.
- ^ Mattesini M., de Almeida J. S., Dubrovinsky L., Dubrovinskaia L., Johansson B., Ahuja R. (2004). "High-pressure and high-temperature synthesis of the cubic TiO2 polymorph". Phys. Rev. B. 70 (21): 212101. Bibcode:2004PhRvB..70u2101M. doi:10.1103/PhysRevB.70.212101.
- ^ a b Dubrovinsky LS, Dubrovinskaia NA, Swamy V, Muscat J, Harrison NM, Ahuja R, Holm B, Johansson B (2001). "Materials science: The hardest known oxide". Nature. 410 (6829): 653–654. Bibcode:2001Natur.410..653D. doi:10.1038/35070650. hdl:10044/1/11018. PMID 11287944. S2CID 4365291.
- ^ Oganov A.R., Lyakhov A.O. (2010). "Towards the theory of hardness of materials". Journal of Superhard Materials. 32 (3): 143–147. arXiv:1009.5477. Bibcode:2010JSMat..32..143O. doi:10.3103/S1063457610030019. S2CID 119280867.
- ^ Al-Khatatbeh, Y., Lee, K. K. M., Kiefer, B. (2009). "High-pressure behavior of TiO2 as determined by experiment and theory". Phys. Rev. B. 79 (13): 134114. Bibcode:2009PhRvB..79m4114A. doi:10.1103/PhysRevB.79.134114.
- ^ Nishio-Hamane D., Shimizu A., Nakahira R., Niwa K., Sano-Furukawa A., Okada T., Yagi T., Kikegawa T. (2010). "The stability and equation of state for the cotunnite phase of TiO2 up to 70 GPa". Phys. Chem. Miner. 37 (3): 129–136. Bibcode:2010PCM....37..129N. doi:10.1007/s00269-009-0316-0. S2CID 95463163.
- ^ Banfield, J. F., Veblen, D. R., Smith, D. J. (1991). "The identification of naturally occurring TiO2 (B) by structure determination using high-resolution electron microscopy, image simulation, and distance–least–squares refinement" (PDF). American Mineralogist. 76: 343.
- ^ "Top 5 Vendors in the Global Titanium Dioxide Market From 2017-2021: Technavio" (Press release). 20 April 2017.
- ^ Hayes T (2011). "Titanium Dioxide: A Shining Future Ahead" (PDF). Euro Pacific Canada. p. 5. Retrieved 16 August 2012.[permanent dead link]
- ^ Hayes (2011), p. 3
- ^ Hayes (2011), p. 4
- ^ "Titanium dioxide". www.essentialchemicalindustry.org.
- ^ Vartiainen J (7 October 1998). "Process for preparing titanium dioxide" (PDF).
- ^ Winkler J (2003). Titanium Dioxide. Hannover: Vincentz Network. pp. 30–31. ISBN 978-3-87870-148-4.
- ^ Withers JC, Cardarelli F, Laughlin J, Loutfy RO. "Recent Improvements for Electrowinning Titanium Metal from Composite Anodes" (PDF). Tucson, AZ: Materials & Electrochemical Research (MER) Corporation.
- ^ Chen, Xiaobo, Mao, Samuel S. (2007). "Titanium Dioxide Nanomaterials: Synthesis, Properties, Modifications, and Applications". Chemical Reviews. 107 (7): 2891–2959. doi:10.1021/cr0500535. PMID 17590053.
- ^ St Clair K (2016). The Secret Lives of Colour. London: John Murray. p. 40. ISBN 978-1-4736-3081-9. OCLC 936144129.
- ^ Hanaor DA, Xu W, Ferry M, Sorrell CC (2012). "Abnormal grain growth of rutile TiO2 induced by ZrSiO4". Journal of Crystal Growth. 359: 83–91. arXiv:1303.2761. Bibcode:2012JCrGr.359...83H. doi:10.1016/j.jcrysgro.2012.08.015. S2CID 94096447.
- ^ Anderson B (1999). Kemira pigments quality titanium dioxide. Savannah, Georgia. p. 39.
{{cite book}}: CS1 maint: location missing publisher (link) - ^ a b Winkler J (2003). Titanium Dioxide. Hannover, Germany: Vincentz Network. p. 5. ISBN 978-3-87870-148-4.
- ^ Margaux de Frouville (28 March 2019). "Deux dentifrices sur trois contiennent du dioxyde de titane, un colorant au possible effet cancérogène" [Two out of three toothpastes contain titanium dioxide, a possibly carcinogenic colouring material] (in French). BFMTV.com.
- ^ State of the Science of Titanium Dioxide (TiO₂) as a Food Additive (PDF). Food Directorate, Health Canada. June 2022. ISBN 978-0-660-44121-4. Archived (PDF) from the original on 30 May 2024.
- ^ "Titanium Dioxide (E171) – Overview, Uses, Side Effects & More". HealthKnight. 10 April 2022. Retrieved 9 June 2022.
- ^ Sherman N (28 May 2025). "Skittles-maker Mars phases out controversial colour additive". BBC News.
- ^ Koleske, J. V. (1995). Paint and Coating Testing Manual. ASTM International. p. 232. ISBN 978-0-8031-2060-0.
- ^ Koleske, J. V. (1995). Paint and Coating Testing Manual. ASTM International. p. 229. ISBN 978-0-8031-2060-0.
- ^ "Pearlescence with Iriodin", pearl-effect.com, archived from the original on 17 January 2012
- ^ Gabros S, Nessel TA, Zito PM (2021), "Sunscreens And Photoprotection", StatPearls, Treasure Island (FL): StatPearls Publishing, PMID 30725849, retrieved 6 March 2021
- ^ Dumbuya H, Grimes PE, Lynch S, Ji K, Brahmachary M, Zheng Q, Bouez C, Wangari-Talbot J (1 July 2020). "Impact of Iron-Oxide Containing Formulations Against Visible Light-Induced Skin Pigmentation in Skin of Color Individuals". Journal of Drugs in Dermatology. 19 (7): 712–717. doi:10.36849/JDD.2020.5032. ISSN 1545-9616. PMID 32726103.
- ^ "US Virgin Islands bans sunscreens harming coral reefs". www.downtoearth.org.in. April 2020. Retrieved 6 March 2021.
- ^ Dan, Yongbo et al. Measurement of Titanium Dioxide Nanoparticles in Sunscreen using Single Particle ICP-MS Archived 6 December 2021 at the Wayback Machine. perkinelmer.com
- ^ "Health_scientific_committees" (PDF).
- ^ a b Jacobs JF, Van De Poel I, Osseweijer P (2010). "Sunscreens with Titanium Dioxide (TiO2) Nano-Particles: A Societal Experiment". Nanoethics. 4 (2): 103–113. doi:10.1007/s11569-010-0090-y. PMC 2933802. PMID 20835397.
- ^ cosmeticsdesign-europe.com (25 September 2013). "Scientists encourage 'safer' rutile form of TiO2 in cosmetics". cosmeticsdesign-europe.com. Retrieved 6 March 2021.
- ^ a b Jaroenworaluck A, Sunsaneeyametha W, Kosachan N, Stevens R (29 March 2006). "Characteristics of silica-coated TiO2 and its UV absorption for sunscreen cosmetic applications". Wiley Analytical Science. 38 (4): 473–477. doi:10.1002/sia.2313. S2CID 97137064 – via Wiley Online Library.
- ^ Dréno B, Alexis A, Chuberre B, Marinovich M (2019). "Safety of titanium dioxide nanoparticles in cosmetics". Journal of the European Academy of Dermatology and Venereology. 33 (S7): 34–46. doi:10.1111/jdv.15943. hdl:2434/705700. ISSN 0926-9959. PMID 31588611. S2CID 203849903.
- ^ a b "OPINION ON additional coatings for Titanium Dioxide (nano form) as UV-filter in dermally applied cosmetic products" (PDF). Scientific Committee on Consumer Safety. European Commission. 7 November 2016 – via ec.europa.eu.
- ^ Wang C, Zuo S, Liu W, Yao C, Li X, Li Z (2016). "Preparation of rutile TiO2@avobenzone composites for the further enhancement of sunscreen performance". RSC Advances. 6 (113): 111865. Bibcode:2016RSCAd...6k1865W. doi:10.1039/C6RA23282E – via Royal society of chemistry.
- ^ Polymers, Light and the Science of TiO2 Archived 29 March 2017 at the Wayback Machine, DuPont, pp. 1–2
- ^ Jorgensen K, Rivkin A, Binzel R, Whitely R, Hergenrother C, Chodas P, Chesley S, Vilas F (May 2003). "Observations of J002E3: Possible Discovery of an Apollo Rocket Body". Bulletin of the American Astronomical Society. 35: 981. Bibcode:2003DPS....35.3602J.
- ^ Aboulouard A, Gultekin B, Can M, Erol M, Jouaiti A, Elhadadi B, Zafer C, Demic S (1 March 2020). "Dye sensitized solar cells based on titanium dioxide nanoparticles synthesized by flame spray pyrolysis and hydrothermal sol-gel methods: a comparative study on photovoltaic performances". Journal of Materials Research and Technology. 9 (2): 1569–1577. doi:10.1016/j.jmrt.2019.11.083. ISSN 2238-7854.
- ^ Mahmoud ZH, Ajaj Y, Kamil Ghadir G, Musaad Al-Tmimi H, Hameed Jasim H, Al-Salih M, Hasen shuhata Alubiady M, Muzahem Al-Ani A, Salih Jumaa S, Azat S, Fadhil Smaisim G, kianfar E (1 January 2024). "Carbon-doped titanium dioxide (TiO2) as Li-ion battery electrode: Synthesis, characterization, and performance". Results in Chemistry. 7: 101422. doi:10.1016/j.rechem.2024.101422. ISSN 2211-7156.
- ^ a b c World Intellectual Property Organization. (2023). "Patent Landscape Report : Production of titanium and titanium dioxide from ilmenite and related applications". www.wipo.int. Patent Landscape Reports. WIPO. doi:10.34667/tind.47029. Retrieved 13 November 2023.
- ^ Liang Chu (2015). "Anatase TiO2 Nanoparticles with Exposed {001} Facets for Efficient Dye-Sensitized Solar Cells". Scientific Reports. 5: 12143. Bibcode:2015NatSR...512143C. doi:10.1038/srep12143. PMC 4507182. PMID 26190140.
- ^ Li Jianming and Dongsheng Xu (2010). "tetragonal faceted-nanorods of anatase TiO2 single crystals with a large percentage of active {100} facets". Chemical Communications. 46 (13): 2301–3. doi:10.1039/b923755k. PMID 20234939.
- ^ M Hussein N Assadi (2016). "The effects of copper doping on photocatalytic activity at (101) planes of anatase TiO 2: A theoretical study". Applied Surface Science. 387: 682–689. arXiv:1811.09157. Bibcode:2016ApSS..387..682A. doi:10.1016/j.apsusc.2016.06.178. S2CID 99834042.
- ^ Hanaor DA, Sorrell CC (2014). "Sand Supported Mixed-Phase TiO2 Photocatalysts for Water Decontamination Applications". Advanced Engineering Materials. 16 (2): 248–254. arXiv:1404.2652. Bibcode:2014arXiv1404.2652H. doi:10.1002/adem.201300259. S2CID 118571942.
- ^ Kurtoglu M. E., Longenbach T., Gogotsi Y. (2011). "Preventing Sodium Poisoning of Photocatalytic TiO2 Films on Glass by Metal Doping". International Journal of Applied Glass Science. 2 (2): 108–116. doi:10.1111/j.2041-1294.2011.00040.x.
- ^ a b c "Discovery and applications of photocatalysis – Creating a comfortable future by making use of light energy". Japan Nanonet Bulletin Issue 44, 12 May 2005.
- ^ Fujishima A, Honda K (1972). "Electrochemical Photolysis of Water at a Semiconductor Electrode". Nature. 238 (5358): 37–8. Bibcode:1972Natur.238...37F. doi:10.1038/238037a0. PMID 12635268. S2CID 4251015.
- ^ "Carbon-doped titanium dioxide is an effective photocatalyst". Advanced Ceramics Report. 1 December 2003. Archived from the original on 4 February 2007.
This carbon-doped titanium dioxide is highly efficient; under artificial visible light, it breaks down chlorophenol five times more efficiently than the nitrogen-doped version.
- ^ Cheap, Clean Ways to Produce Hydrogen for Use in Fuel Cells? A Dash of Disorder Yields a Very Efficient Photocatalyst. Sciencedaily (28 January 2011)
- ^ Karvinen S (2003). "Preparation and Characterization of Mesoporous Visible-Light-Active Anatase". Solid State Sciences. 5 2003 (8): 1159–1166. Bibcode:2003SSSci...5.1159K. doi:10.1016/S1293-2558(03)00147-X.
- ^ Bian L, Song M, Zhou T, Zhao X, Dai Q (June 2009). "Band gap calculation and photo catalytic activity of rare earths doped rutile TiO2". Journal of Rare Earths. 27 (3): 461–468. doi:10.1016/S1002-0721(08)60270-7.
- ^ Advanced Concrete Pavement materials Archived 20 June 2013 at the Wayback Machine, National Concrete Pavement Technology Center, Iowa State University, p. 435.
- ^ Hogan, Jenny (4 February 2004) "Smog-busting paint soaks up noxious gases". New Scientist.
- ^ TIME's Best Inventions of 2008. (31 October 2008).
- ^ Winkler J (2003). Titanium Dioxide. Hannover: Vincentz Network. pp. 115–116. ISBN 978-3-87870-148-4.
- ^ Konstantinou IK, Albanis TA (2004). "TiO2-assisted photocatalytic degradation of azo dyes in aqueous solution: Kinetic and mechanistic investigations". Applied Catalysis B: Environmental. 49 (1): 1–14. Bibcode:2004AppCB..49....1K. doi:10.1016/j.apcatb.2003.11.010.
- ^ Hanaor DA, Sorrell CC (2014). "Sand Supported Mixed-Phase TiO2 Photocatalysts for Water Decontamination Applications". Advanced Engineering Materials. 16 (2): 248–254. arXiv:1404.2652. doi:10.1002/adem.201300259. S2CID 118571942.
- ^ Ramsden JJ (2015). "Photocatalytic antimicrobial coatings". Nanotechnology Perceptions. 11 (3): 146–168. doi:10.4024/N12RA15A.ntp.15.03.
- ^ Jones T, Egerton TA (2000). "Titanium Compounds, Inorganic". Kirk-Othmer Encyclopedia of Chemical Technology. John Wiley & Sons, Inc. doi:10.1002/0471238961.0914151805070518.a01.pub3. ISBN 978-0-471-23896-6.
- ^ a b c Hirakawa T, Nosaka Y (23 January 2002). "Properties of O2•-and OH• formed in TiO2 aqueous suspensions by photocatalytic reaction and the influence of H2O2 and some ions". Langmuir. 18 (8): 3247–3254. doi:10.1021/la015685a.
- ^ Mogilevsky G, Chen Q, Kleinhammes A, Wu Y (2008). "The structure of multilayered titania nanotubes based on delaminated anatase". Chemical Physics Letters. 460 (4–6): 517–520. Bibcode:2008CPL...460..517M. doi:10.1016/j.cplett.2008.06.063.
- ^ a b Wang, Cui (2015). "Hard-templating of chiral TiO2 nanofibres with electron transition-based optical activity". Science and Technology of Advanced Materials. 16 (5): 054206. Bibcode:2015STAdM..16e4206W. doi:10.1088/1468-6996/16/5/054206. PMC 5070021. PMID 27877835.
- ^ Wu Y (1 January 2007), Wu Y (ed.), "15 - PREPARATION OF ULTRAFINE POWDERS BY REACTION–PRECIPITATION IN IMPINGING STREAMS III: NANO TITANIA", Impinging Streams, Amsterdam: Elsevier Science B.V., pp. 301–315, ISBN 978-0-444-53037-0, retrieved 15 November 2024
- ^ a b Warheit DB, Donner EM (November 2015). "Risk assessment strategies for nanoscale and fine-sized titanium dioxide particles: Recognizing hazard and exposure issues". Food Chem Toxicol (Review). 85: 138–47. doi:10.1016/j.fct.2015.07.001. PMID 26362081.
- ^ a b c "Titanium Dioxide as a Color Additive in Foods". US Food and Drug Administration. 4 March 2024. Archived from the original on 15 March 2024.
- ^ Warheit DB (2024). "Safety of titanium dioxide (E171) as a food additive for humans". Frontiers in Toxicology. 6: 1333746. doi:10.3389/ftox.2024.1333746. ISSN 2673-3080. PMC 11295244. PMID 39100893.
- ^ Kirkland D, Aardema MJ, Battersby RV, et al. (1 December 2022). "A weight of evidence review of the genotoxicity of titanium dioxide (TiO2)". Regulatory Toxicology and Pharmacology. 136: 105263. doi:10.1016/j.yrtph.2022.105263. ISSN 0273-2300. PMID 36228836.
- ^ "France to ban titanium dioxide whitener in food from 2020". Reuters. 17 April 2019.
- ^ Boffey D (6 May 2021). "E171: EU watchdog says food colouring widely used in UK is unsafe". the Guardian.
- ^ "UK disagrees with EU position on titanium dioxide". Food Safety News. 9 March 2022.
- ^ "Titanium dioxide (TiO2) as a food additive". Health Canada. 6 April 2023.
- ^ 'amending Annexes II and III to Regulation (EC) No 1333/2008 of the European Parliament and of the Council as regards the food additive titanium dioxide (E 171)'. Commission Regulation (EU) 2022/63, 14 January 2022
- ^ Smith DG (13 April 2023). "Two States Have Proposed Bans on Common Food Additives Linked to Health Concerns". The New York Times. Archived from the original on 13 November 2023. Retrieved 15 November 2023.
- ^ Bedigan M (12 June 2024). "Scientists raise alarm over sunscreen ingredient being found in cakes and candies". The Independent. Retrieved 13 June 2024.
- ^ "Dunkin' Donuts to remove titanium dioxide from donuts". CNN Money. March 2015.
- ^ Winkler HC, Notter T, Meyer U, Naegeli H (December 2018). "Critical review of the safety assessment of titanium dioxide additives in food". Journal of Nanobiotechnology. 16 (1): 51. doi:10.1186/s12951-018-0376-8. ISSN 1477-3155. PMC 5984422. PMID 29859103.
- ^ Titanium dioxide (PDF). Vol. 93. International Agency for Research on Cancer. 2006.
- ^ "Titanium Dioxide Classified as Possibly Carcinogenic to Humans". Canadian Centre for Occupational Health & Safety. August 2006.
- ^ National Institute for Occupational Safety and Health. "Current Intelligence Bulletin 63: Occupational Exposure to Titanium Dioxide (NIOSH Publication No. 2011-160)" (PDF). National Institute for Occupational Safety and Health.
- ^ a b c Tourinho PS, van Gestel CA, Lofts S, Svendsen C, Soares AM, Loureiro S (1 August 2012). "Metal-based nanoparticles in soil: Fate, behavior, and effects on soil invertebrates". Environmental Toxicology and Chemistry. 31 (8): 1679–1692. Bibcode:2012EnvTC..31.1679T. doi:10.1002/etc.1880. ISSN 1552-8618. PMID 22573562. S2CID 45296995.
- ^ Swiler DR (2005). "Pigments, Inorganic". Kirk-Othmer Encyclopedia of Chemical Technology. John Wiley & Sons, Inc. doi:10.1002/0471238961.0914151814152215.a01.pub2. ISBN 978-0-471-23896-6.
- ^ Preočanin T, Kallay N (2006). "Point of Zero Charge and Surface Charge Density of TiO2 in Aqueous Electrolyte Solution as Obtained by Potentiometric Mass Titration". Croatica Chemica Acta. 79 (1): 95–106. ISSN 0011-1643.
External links
[edit]- International Chemical Safety Card 0338
- NIOSH Pocket Guide to Chemical Hazards
- "Titanium Dioxide Classified as Possibly Carcinogenic to Humans", Canadian Centre for Occupational Health and Safety, August, 2006 (if inhaled as a powder)
- A description of TiO2 photocatalysis
- Titanium and titanium dioxide production data (US and World)


 French
French Deutsch
Deutsch
![SEM (top) and TEM (bottom) images of chiral TiO2 nanofibers[84]](http://upload.wikimedia.org/wikipedia/commons/thumb/b/b5/Chiral_TiO2_nanofibers_2.jpg/120px-Chiral_TiO2_nanofibers_2.jpg)


