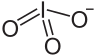
碘酸铪是一种无机化合物,化学式为Hf(IO3)4,它可由水合氧化铪和五氧化二碘的水溶液(碘酸)反应得到,根据反应条件不同还可以得到HHf(IO3)5·3H2O。[2]Hf(IO3)4难溶于水[3]和酸[4]。
参考文献[编辑]
- ^ Christo Balarew; E. V. Zagnit’ko; Jitka Eysseltova; Jean-Jacques Counioux. IUPAC-NIST Solubility Data Series. 85. Transition and 12–14 Main Group Metals, Lanthanide, Actinide, and Ammonium Halates. J. Phys. Chem. Ref. Data 37, 933–1118 (2008). doi:10.1063/1.2804088.
- ^ Deabriges, Jean; Rohmer, Raymond. Reactions of aqueous iodic acid solutions on hydrated zirconium and hafnium oxides(法文). Bulletin de la Societe Chimique de France, 1968. 2: 521-527. ISSN: 0037-8968.
- ^ Hiroshi Miyamoto, et al. IUPAC–NIST Solubility Data Series #. Transition and 12 to 14 Main Group Metals, Lanthanide, Actinide and Ammonium Halates. st.hirosaki-u.ac.jp. (pdf (页面存档备份,存于互联网档案馆))
- ^ Fetto Pasquale, et al. La Chimica nella Scuola (pdf (页面存档备份,存于互联网档案馆)). Societa Chimica Italiana.


 French
French Deutsch
Deutsch